
| Welcome to the Club of Amsterdam Journal. “Biomedicine is booming. Recent advances in Genomics, Systems Biology, Nanomedicine, Tissue Engineering, Biophotonics and other disciplines have not only raised hopes for new drugs and diagnostics, but have also made it more likely than ever that real innovative products for improving Human Health will be available within 3-8 years. The announcement on March 6 of this year that US president Obama ends Bush’s ban on embryo stem cell research will allow US researchers to catch up with their EU and Far-East colleagues, who were restricted in finding good stem cell sources for purposes of Tissue Engineering. Microarray technology makes it possible and feasible to quickly monitor thousands of potentially active compounds in their ability to bind to receptors or affect cell metabolism, leading to new drugs. Through genomics the expression of countless genes can be measured and the results can be used to develop personalized medicine, probiotics or functional foods.Biophotonics has become an extremely sensitive tool to support medical imaging on a molecular scale, leading to earlier diagnostics in cancer research, allowing for earlier intervention. The presentations will focus on the above developments and their role in maintaining and improving human health.” Event concept: Gerhard Mulder, Senior Consultant, Syntens In case you would like to hear more and also share your thoughts, then email us or visit our next event about the future of BioMed on May 28. Location : Syntens, De Ruyterkade 5, 1013 AA Amsterdam And a special announcement: The Club of Amsterdam visits the RSA in London. the future of Connectivity, Thursday, June 25, 2009 Felix Bopp, editor-in-chief |
Healthcare Manifesto
Quotes from the Healthcare Manifesto by EuropaBio – the European Association for Bioindustries
“Healthcare biotechnology refers to a medicinal or diagnostic product or a vaccine that consists of, or has been produced in, living organisms and may be manufactured via recombinant technology. This technology has a tremendous impact on meeting the needs of patients and their families – as it not only encompasses medicines and diagnostics that are manufactured using a biotechnological process, but also gene and cell therapies and tissue engineered products.
Today, the majority of innovative medicines, whether manufactured using biotechnology or via a chemical synthesis like a traditional small molecule medicine, and many diagnostic products, are made available by applying modern biotechnology in their development and/or manufacturing processes.”
[…]
Did you know that
- More than 350 million patients have benefited from approved medicines manufactured through biotechnology and gene technology to treat or prevent heart attacks, stroke, multiple sclerosis, breast cancer, cystic fibrosis, leukaemia, hepatitis, diabetes and other diseases, including rare diseases.
- Today, more than 600 new biotech medicines and vaccines are being tested for more than 100 diseases pushing the frontiers of science. Biotech medicines are estimated to account for more than 20% of all marketed medicines and about half of all medicines in the pipeline.
- The industry in Europe in 2007 comprised more than 1,700 companies and represented a market worth more than €17 billion.
- Healthcare biotechnology continues to grow annually at an average rate of 20% (more than double that of traditional pharma) and that it is 7 times larger than it was 10 years ago.
- Currently, the leading classes of biotech therapies are growth factors for blood cells (used in treating anemia resulting from chronic kidney disease, from chemotherapy and radiation treatments, and from other critical illnesses such as heart failure); cancer treatments (new and targeted treatments for cancers – such as the use of monoclonal antibodies), treatment of autoimmune diseases, enzyme replacement therapies to treat rare diseases and anti-diabetic therapies.
- For the first time in the history of human healthcare, biotechnology is enabling the development and manufacturing of therapies for a number of rare and very rare genetic diseases, collectively affecting some 20 to 30 million Europeans and their families.
- Biotechnology has a major impact on the provision of safe and effective vaccines against infectious diseases, and provides safer recombinant alternatives to proteins derived from human blood or tissue.
[…]
Towards Patient-Centred Healthcare Systems
In 2004, the European Commission ambitiously launched a series of reforms at national and European level aimed at making the European Union (EU) “the most dynamic and competitive knowledge based economy in the world” by 2010.
To support this, biotechnology and healthcare became a high priority on the political agenda of both the Member States and of the European Union. Innovation in these two areas been and will continue to be a key element in achieving this goal. However,
policies in these two areas need to become more interlinked, as practically all future medicines and therapies will use healthcare biotechnology in the R&D process and/or in manufacturing.
EuropaBio calls for healthcare systems in Europe that are committed to put the patient at the centre of any medical, social, economical and ethical consideration. Budgetary considerations should be secondary to this overarching political mandate and its
implementation.
Designing policy to foster innovation in biotechnology is not an easy task, however; biotechnology is a complex field with complex potential outcomes and impacts while new insights need to be thoroughly discussed with and within society. The policy-making process faces a number of further challenges when transferred to the healthcare sector – including a small knowledge base mostly found within the companies themselves, complex ideas to pass on to patients, new approaches to promote development of treatments for unmet but rare medical needs, treating diseases that introduce new concepts and paradigms into patient/physician relationships (e.g.: monoclonal antibodies and personalised medicine), a significant SME (Small and Medium-Size Enterprises) base, and potentially polarising issues (such as some stem cell-based therapies).
The EU addresses this issue through a range of initiatives such as the introduction of the orphan medicinal products regulation to foster development of medicines to treat rare diseases, opening an SME office at the EMEA and regulating advanced therapies
(cell, gene and tissue therapies).
EuropaBio was a full member of the High Level Pharmaceutical Forum for its expert input in these areas. 2009/10 will see a continuation of these initiatives. Building on the EuropaBio Healthcare Vision, this Manifesto sets out how EuropaBio will contribute to this process of policy development, as well as regulatory implementation, in 2009/10. These actions are laid out in the Healthcare Council Mission, four areas that will need to be addressed in order to handle the healthcare innovations expected over the next few years that will provide answers to unmet medical needs.
[…]
Biotechnology: The Invisible Revolution
by EuropaBio
Next Event

the future of BioMed
Thursday, May 28, 2009
Registration: 18:30-19:00, Conference: 19:00-21:15
Location: Syntens, De Ruyterkade 5, 1013 AA Amsterdam
The speakers and topics are
Arjen Brinkman, Director, Personal Space Technologies
Using Virtual Reality (VR) to improve human health
Michael Münker, STEP 2 B.V.
Inventions to Innovation: Lessons from Medical Devices
Jeanine van de Wiel, Global Regulatory Affairs Manager, DSM Food Specialties
Personalized Nutrition, an emerging business area
Our moderator is Gerhard Mulder, Senior Consultant, Syntens
Club of Amsterdam blog

News about the Future

European Institute of Innovation and Technology
The European Institute of Innovation and Technology (EIT) is to be a key driver of sustainable European growth and competitiveness through the stimulation of world-leading innovations with a positive impact on economy and society.
The mission of the EIT is to grow and capitalise on the innovation capacity and capability of actors from higher education, research, business and entrepreneurship from the EU and beyond through the creation of highly integrated Knowledge and Innovation Communities (KICs).

Toys Guide
WECF- Women in Europe for a Common Future – has published a Toys Guide which provides tips on choosing toxic- free toys. The practical brochure provides concise information about the most hazardous substances found in toys and what the potential dangers are, and provides concrete tips on how consumers can play it safe when buying toys for children.
“Parampara” (Tradition) Project

Arnab B. Chowdhury is founder and CEO of Ninad ~ an e-Learning consulting and collaborating network that participated in the “Parampara” project. Ninad offers consulting services that are centered in Integrality.

A case study
A poet once said that memory is man’s real possession; in nothing else is mankind so rich, or so poor. In the confines of the Institut Français de Pondicherry (IFP, www.ifpindia.org) and the Ecole Française d’Extrême-Orient (EFEO), two French governmental research institutes, Indian and French researchers have been working hand-in-hand for over 30 years on a fascinating undertaking to create a database of one of the world’s richest collection of manuscripts devoted to the “Saivasiddhanta”, a Saiva religio-philosophical system written in verse – a prominent aspect of Hinduism. They are ensuring that India conserves a vital component of her rich cultural memory via e-Culture while synergising appropriate cultural pedagogy, knowledge management, communication design with smart ICT.
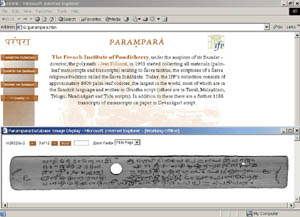
Project: The manuscript collection of the IFP was initiated in 1955 under the auspices of IFP founder-director, Jean Filliozat. Bundles were brought back to the IFP from the private collections of priests and temples across South India. When the manuscripts themselves could not be obtained, transcripts in Devanagiri script (as in Hindi) were made. More than half the collection consists of Saiva manuscripts, comprising:
- Approximately 60,000 texts preserved in 8,600 palm-leaf bundles.
- 1,144 transcripts of manuscripts on paper.
- Precisely 6,850 are in Sanskrit (of which approximately 60% are in Grantha script).
- 1,200 in Tamil or Tamil and Sanskrit.
- A few are in Tulu, Telugu, Malayalam and Kannada (regional Indian languages).
In addition to the Saiva materials, the wide range of subjects covered includes Astrology, Puranas (Mythology), Siddha Medicine, Veda, Epics, Belles Lettres and Tamil devotional literature.
Process: The digitisation and transliteration project, aptly named “Parampara” (denoting ‘tradition’ in Sanskrit), got into full swing in 1997 and the entire collection is presently housed and being painstakingly catalogued at both the research institutes in air-conditioned conservation-chambers. Its first CDROM release is of a unique digital archiving system, published under the name “Parampara”, co-produced by the IFP, EFEO and the Chennai-based AMM Foundation with a multi-lateral team of Indology scholars, media experts and information technologists. Currently a collaborative Indo-French project has been framed with National Mission for Manuscripts initiated by the Ministry of Tourism and Culture, Government of India.
Challenge: The cataloguing process is a challenge on its own since each bundle of leaves may contain dozens of texts, and there are no headings, no word-breaks and no rubrication of titles or colophons. This requires not only a sound knowledge of the Saivasiddhanta system but also the ability to read handwritten scripts, in particular Grantha, a dying skill in Tamil Nadu (southern Indian state) today. Each folio is studied and the texts are identified. A descriptive form is filled out that describes each manuscript along with an English transliteration. These details are then entered into a database.
As far as e-Culture is concerned, the challenge lay in reflecting the taxonomy of how the palm-leaf manuscripts and their content have been collated over millennia (6th century A.D onwards) onto a suitable taxonomy of a graphics-based database that is scalable with efficient access.
In recent times, thanks to the help of the Muktabodha Indological Research Institute (India), a major part of the collection (namely all the transcripts) can now be downloaded online free of charge: (http://muktalib.org/access_page.htm)
Technology: From a software perspective, all images of manuscripts and books in the online digital library are made available in two formats, DjVu (http://djvu.org) and PDF.
The DjVu format is ideal for the viewing of large books and manuscripts on the web and uses an encoding scheme called “wavelet” technology. Essentially this means that they are stored as mathematical formulas describing curves. This allows the page images to be zoomed with almost no loss of quality to 1200% and without getting the jagged edges (called “jaggies”) that occur if the image is stored as a pattern of pixels.
The PDF format is ideal for the downloading and disseminating of files of books or manuscripts because it is so universally available and known. The online database of the 210,000 pages of transcripts occupies 102 Gigabytes of storage and is hosted on a Linux Apache server. Additionally, this e-Culture application applies Open Source technologies such as PHP and MySQL.
Conclusion: In all, we can see how ICT with a sincere pedagogical sensitivity towards culture, can help unravel and disseminate the cultural and research Knowledge-Value from the past to make it accessible, comprehensive; to transport it from the past to the present and ensure its future.
Appreciation: The “Parampara” project is now a part of UNESCO’s ‘Memory of the World’ program – an initiative to recognize the immense cultural significance of various sites worldwide.
NASA Leads Team in Establishing a Renewable Hydrogen

NASA’s Glenn Research Center is leading a team of industry and university partners in demonstrating a prototype of a commercial hydrogen fueling station that uses wind and solar power to produce hydrogen from water. This initial installation will produce hydrogen from Lake Erie water to fuel a mass transit bus powered by fuel cells.
The demonstration, featuring a unique, high-capacity electrolyzer that separates water into its elemental components of hydrogen and oxygen, is part of an economic development program in the Cleveland area. Local workers will design and build the electrolyzer using commercially available components.
The Glenn-led collaboration will customize the electrolyzer for the prototype fueling station, and design the circuitry needed to use renewable energy sources to power the electrolyzer and fueling station.
“The project is more than a key technology demonstration,” said project team member Valerie Lyons, chief of Glenn’s Power and In-Space Propulsion Division. “It will be a great educational tool for the public and will serve as a catalyst to inspire new ideas and initiatives that can generate many new jobs and manufacturing opportunities in Ohio.”
The hydrogen fueling station will be located in downtown Cleveland at the Great Lakes Science Center on the south shore of Lake Erie, where it can be powered from the science center’s existing wind and solar power sources. The fueling station will generate hydrogen from Lake Erie water for use in a Greater Cleveland Regional Transit Authority bus powered by fuel cells. The transit authority will operate the bus in revenue service.
Cleveland State University’s Nance College of Business Administration will work alongside the collaborators to develop a business template for the electrolyzer and station. The designs for both will be treated as intellectual property and placed in a trust benefiting Ohio citizens.
The build-up of the electrolyzer, a major step toward the reality of the fueling station, is funded by the Ohio Aerospace Institute through a $310,000 grant from The Cleveland Foundation. The initial funding is $110,000, with an additional $200,000 to be provided for milestone progress.
The goals of the economic development program include engaging Ohio’s supply chain manufacturers and retraining a skilled work force for clean energy jobs. The project will demonstrate the viability of clean energy systems for transportation and stationary power and boost regional economic development.
Other collaborators include Hydrogen and Fuel Cell Consultants of Brecksville, Ohio; the Center for Automotive Research at Ohio State University in Columbus, Ohio; Parker Hannifin and Technology Management, Inc. of Cleveland; Sierra Lobo of Milan, Ohio; Hamilton Sundstrand of Windsor Locks, Conn.; the University of Toledo; and the Earth Day Coalition of Cleveland.
Special Announcement: the future of Connectivity

the future of Connectivity
The Club of Amsterdam visits the RSA in London
Thursday, June 25, 2009
Receptionn: 18:30-19:00, Conference: 19:00-21:15
Location: Royal Society for the encouragement of Arts, Manufactures and Commerce (RSA), 8 John Adam Street, London WC2N 6EZ
Supporters: LogMeIn, RSA
Egbert-Jan Sol, CTO, TNO Science and Industry, co-founder/1st chairman, DSE
Peter Cochrane, Co-Founder, Cochrane Associates
Evolving Connectivity
Hardy F. Schloer, Owner, Schloer Consulting Group
Human Connectivity at the Event Horizon of new AI Technologies
Moderated by James Cridland, Head of Future Media & Technology, BBC Audio & Music Interactive
Recommended Book

Beyond Borders: global biotechnology report 2008
This2008’s “Beyond Borders” report by Ernst & Young highlights a biotechnology industry that continues to grow rapidly, as companies raise record amounts of venture capital and the potential value of mergers, acquisitions and alliances reaches new heights.
Futurist Portrait: Don Tapscott

Don Tapscott (born 1947) is a Canadian business executive, author, consultant and speaker based in Toronto, Ontario, specializing in business strategy, organizational transformation and the role of technology in business and society. Tapscott is chairman of business strategy think tank New Paradigm (now nGenera), which he founded in 1993. Tapscott is also Adjunct Professor of Management at the Joseph L. Rotman School of Management, University of Toronto.
Tapscott holds a B.Sc. in Psychology and Statistics, and an M.Ed. specializing in Research Methodology. He also holds two honorary Doctor of Laws (honoris causa) granted by the University of Alberta in 2001, and Trent University in 2006] While earning his Master’s of Education at the University of Alberta, he ran for mayor of Edmonton in the 1977 municipal election.
Tapscott has authored or co-authored eleven books on the application of technology in business and society. His most famous book, co-authored with Anthony Williams and published in 2006 is Wikinomics: How Mass Collaboration Changes Everything (with an expanded edition to be published in April 2008). His penultimate book, co-authored with David Ticoll in 2003 is The Naked Corporation: How the Age of Transparency Will Revolutionize Business. The Naked Corporation describes how corporate transparency, accountability, and stakeholder relationships are the new frontier for competitive innovation.
He has also co-authored, Digital Capital: Harnessing the Power of Business Webs. This book describes how business webs are replacing the traditional model of the firm and changing the dynamics of wealth creation and competition. His popular 1997 book Growing Up Digital: The Rise of the Net Generation explained the business and social impact of the first generation to come of age in the digital age. Digital Economy: Promise and Peril In The Age of Networked Intelligence, published in 1995, was one of the first books to describe how the Internet would change business and society. Tapscott is probably best known for his 1992 book Paradigm Shift: The New Promise of Information Technology.
Don Tapscott talks about how the New Generation is changing the world by developing new ways of thinking and interacting. His latest book is called “Grown Up Digital”.
Agenda
  Our Season Program 2008 / 2009:  The Club of Amsterdam visits the RSA in London .June 25. 2009 the future of Connectivity Location: Royal Society for the encouragement of Arts, Manufactures and Commerce (RSA), 8 John Adam Street, London WC2N 6EZ |




Customer Reviews
Thanks for submitting your comment!